FDA: Global Pharma's eye drops contaminated with “filth” while made unapproved claims for eye drops

Viral Kana Po

Regulatory Affairs Professionals Society (RAPS) on LinkedIn: FDA blasts California firm for multiple GLP issues involving nonclinical…

FDA Investigations Operations Manual - Public Health Information

Pharma Manager`s Association

Anger, Faith, and Growth
Witch's Pumpkin (Airdrop) - 🔥🔥 Check full Collection for other Amazing NFTs 🔥🔥 - NFTNAMA

Regulatory Affairs Professionals Society (RAPS) on LinkedIn: FDA: Global Pharma's eye drops contaminated with “filth” while made…

Hearts of Oak Podcast – Podcast – Podtail
What is an example of a long term problem that has been solved by technology? - Quora

Forensic Pharmacy Complete Notes

OGPS Update newsletter - January 2023

Afrilabs – Visa Foundation: $100,000 USD for women entrepreneurs

Cosmeceuticals

Orion Magazine - Forget Shorter Showers
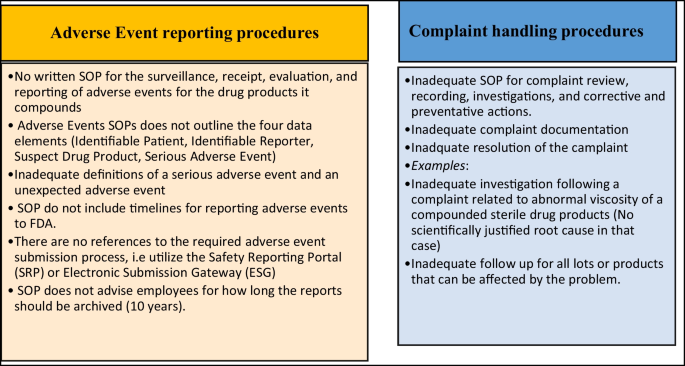
Content Analysis of US FDA Warning Letters Issued to Compounding Pharmacies Regarding Violations of Current Good Manufacturing Practices Between 2017 and 2022